| Home | Energy | Nuclear Fusion | Electricity | Climate Change | Lighting Control | Contacts | Links |
|---|
XYLENE POWER LTD.
CARBON POOLS:
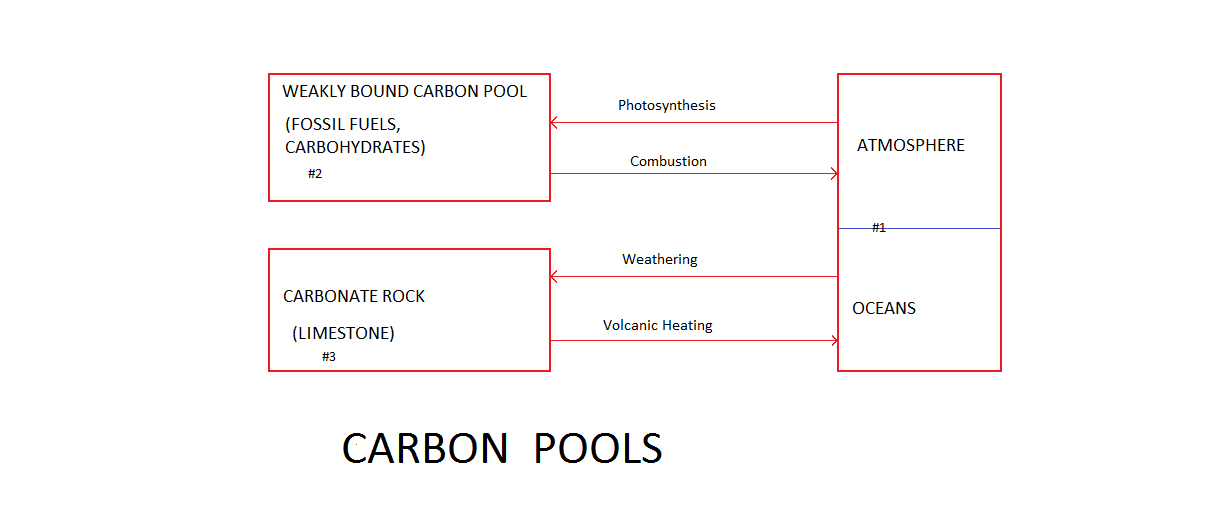
Carbon is a stable element. The total amount of carbon on Earth has remained almost constant for billions of years. Most of the carbon atoms near the Earth's surface are in one of three pools.
Pool #1, also known as the ocean-atmosphere pool, includes: carbon dioxide (CO2) gas in the air and CO2 dissolved in the oceans as H2CO3, and existing in the ocean as half of the bicarbonate (HCO3)- ion concentration. Note that the other half of the bicarbonate ion concentration is assigned to Pool #3 as it is part of the carbonate rock and never converts to gaseous CO2;
Pool #2 includes various forms of free and weakly chemically bound carbon including: biochar, graphite, diamond, coal, natural gas, frozen methane hydrate (CH4.5.75H2O), petroleum and biomatter carbohydrates such as (C6H12O6);
Pool #3 is composed of metal carbonate rocks such as limestone (CaCO3).

INTER-POOL CARBON TRANSFERS:
When the Earth's surface was very hot there was almost no carbon in Pool #3 and all the carbon in Pool #1 was in the atmosphere.
Over very long periods of time at normal temperatures solar driven biological processes move carbon from Pool #1 to Pool #2.
Over very long periods of time at normal temperatures weathering of silicate rocks moves carbon from Pool #1 to Pool #3.
At steady state conditions natural processes transfer CO2 from the air to bicarbonate (HCO3)- ions dissolved in the oceans or vice versa with a time constant of about 16 years, whereas natural processes that move carbon from Pool #1 to Pool #2 via formation of fossil fuels or from Pool #1 to Pool #3 via formation of metal carbonate rocks have a time constant of about 200,000 years.
Combustion rapidly transfers carbon from Pool #2 to Pool #1. Similarly an increase in temperature that melts methane hydrate rapidly transfers carbon from Pool #2 to Pool #1. In the atmosphere CH4 spontaneously reacts with O2 within a decade to produce CO2 + H2O. Humans are making the Earth uninhabitable by transferring massive amounts of carbon from Pool #2 to Pool #1 via combustion of fossil fuels.
Direct contact between hot volcanic lava and metal carbonate rocks on the Earth's surface heats the metal carbonate rocks sufficiently to rapidly move carbon from Pool #3 to Pool #1.
Life as we know it is only possible when the amount of CO2 in Pool #1 is within an appropriate range. Too much CO2 in Pool #1 is toxic to animal respiration and makes the Earth too hot. Too little CO2 in Pool #1 prevents plant respiration and makes the Earth too cold.
EARTH HISTORY:
Early in the Earth's history the Earth's atmosphere was thick, rich in CO2 and water (H2O) vapor, and contained no free oxygen ((O2)). The Earth's surface gradually cooled by emission of thermal radiation. Part of the atmospheric H2O vapor condensed to form liquid water. Part of the atmospheric CO2 dissolved in the liquid H2O to form carbonic acid ((H2CO3)) which very slowly converted near surface volcanic metal silicate rocks such as calcium silicate ((CaSiO3)) into metal carbonate rocks such as limestone ((CaCO3)) plus silica ((SiO2)) sand, in accordance with the chemical equation:
CaSiO3 + CO2 = CaCO3 + SiO2.
The average ocean water temperature was much warmer than today due to a higher atmospheric CO2 concentration and a lower planetary albedo. Due to the higher average ocean water temperature the ratio of CO2 in the atmosphere to CO2 in dissolved bicarbonate ((HCO3)-) ions in the ocean was larger than today. The total amount of CO2 in the ocean-atmsophere pool was much larger than today.
Then a biological process known as photosynthesis commenced. For many millions of years the flow of radiant energy from the sun in combination with plant photosynthesis and subsurface anaerobic biochemical reactions caused a gradual transfer of carbon from the ocean-atmosphere pool (Pool #1) to Pool #2. This carbon transfer process raised the atmospheric oxygen ((O2)) concentration from 0% to about 20% and lowered the atmospheric carbon dioxide (CO2) concentration to about 1%. Meanwhile absorption of CO2 by silicate rock to form carbonate rock continued.
The reduced atmospheric CO2 concentration allowed increased infrared emission by the Earth, which reduced the atmospheric temperature. This reduced atmospheric temperature gradually cooled the deep ocean which allowed the ocean to absorb more atmospheric CO2 by conversion of exposed insoluble marine metal carbonate rock such as CaCO3 into water soluble metal ions such as Ca++ and bicarbonate (HCO3)- ions, in accordance with the chemical equation:
CO2 + H2O + CaCO3 = Ca++ + 2 (HCO3)-
By about 56 million years ago these processes had left the Earth with an atmosphere containing about 21% oxygen and about 500 ppmv of CO2 and the oceans with about twice the (HCO3)- ion concentration as today. Then came the Paleocene Eocene Thermal Maximum (PETM) that rapidly transferred sufficient carbon from Pool #2 to Pool #1 to increase Pool #1 by about 18%. This rapid carbon transfer caused sufficient atmospheric heating to cause thermal runaway and trap the Earth in its warm state. Then ongoing formation of carbonate rocks and fossil fuels gradually extracted CO2 from Pool #1, which after about 200,000 years reduced the atmospheric CO2 concentration below 626 ppmv, which stopped thermal runaway and enabled commencement of ocean cooling. After a further 300,000 years the amount of carbon in Pool #1 decreased the steady state atmospheric CO2 concentration to about 500 ppmv, the level prior to the PETM.
Ongoing formation of carbonate rocks and fossil fuels over the 55 million years between the PETM and the beginning of the industrial revolution further reduced the steady state atmospheric CO2 concentration to about 280 ppmv.
In regions where the temperature is consistently below 0 degrees C methane that previously formed as a result of anaerobic biological processes became trapped in a frozen methane hydrate CH4.5.75H2O complex known as a clathrate.
As a result of gradual natural extraction of carbon from Pool #1 the Earth further cooled, causing formation of land borne glaciers that lowered the average sea level. The combination of a high atmospheric oxygen concentration, a low atmospheric carbon dioxide concentration, moderate temperatures, a reduced sea level and land borne glaciers/snowpacks has persisted for several million years and has enabled the evolution of modern animals, including human beings. Some of the present south polar ice cap has been in place for over 400,000 years. Evidence of repeated glaciations is apparent in ocean sediment drill cores going back 2.5 million years.
Prior to the industrial revolution the Earth's atmospheric CO2 concentration was maintained by an approximate steady state balance between the low latitude rate of CO2 emission by the ocean due to solar powered evaporation of ocean foam containing bicarbonate (HCO3)- ions and the high latitude rate of CO2 absorption by the ocean due to a higher CO2 solubility at low ocean temperatures. Note that the rate of CO2 absorption by the ocean is also proportional to the high latitude exposed open ocean area which changes with ice cover.
ICE AGES:
When the Arctic Ocean forms an ice cover the high latitude exposed open ocean area decreases which reduces the rate of CO2 absorption by the oceans. As a consequence the atmospheric CO2 concentration gradually rises by about 30 ppmv at which point CO2 related global warming causes the Arctic Ocean ice to melt.
When the Arctic Ocean ice melts the high latitude exposed open ocean area increases which increases the rate of CO2 absorption by the oceans. Then the atmospheric CO2 concentration gradually falls about 30 ppmv at which point global cooling causes the Arctic Ocean to form a new ice cover.
Ice core data shows that prior to the industrial revolution the Earth's atmosphere went through over 30 such atmospheric CO2 concentration cycles. The typical cycle period was about 12,000 years. These cycles are known as ice ages.
Ice core data also shows that superimposed on the 12,000 year atmospheric CO2 concentration cycles is a longer term oscillation with a period of about 120,000 years and a peak to peak change in CO2 concentration of about 90 ppmv. This longer period oscillation is believed to be due to an oscillation in deep ocean temperature which might be related to an astrophysical effect that causes a long term oscillation in solar irradiance.
Ice core data shows that the combination of these two oscillatory effects resulted in the atmospheric CO2 concentration varying between 180 ppmv and 300 ppmv over the 400,000 years preceeding the industrial revolution.
FOSSIL FUEL FORMATION:
During periods of moderate temperature forests grow causing accumulation of biomass (wood) on the Earth's surface. Most of the forest biomass consists of carbohydrate isomers that have the approximate chemical formula C6H12O6. When ice ages occur or when volcanos errupt, part of the accumulated biomass may be trapped in anaerobic (oxygen free) conditions due to being covered by ice, water, silt, volcanic ash or lava. In anaerobic conditions carbohydrates decompose to form fossil fuels.
Major fossil fuel deposits take millions of years to form because the conditions for burial and anaerobic decomposition of large amounts of biomatter are relatively infrequent. The existing deep fossil fuel reserves on Earth are the result of billions of years of natural fossil fuel accumulation. However today mankind is rapidly converting these natural fossil fuel reserves into atmospheric CO2.
COAL:
At temperatures above 60 degrees C the dominant anaerobic carbohydrate decomposition reaction is:
C6H12O6 = 6 C + 6 H2O
Lava, which is primarily dehydrated silicate rock, will absorb water (H2O) of hydration, leaving behind coal (C).
NATURAL GAS:
At temperatures in the range 30 degrees C to 50 degrees C there are multiple natural anaerobic biochemical carbohydrate decomposition reactions of the form:
C6H12O6 = 3 CH4 + 3 CO2
The released carbon dioxide (CO2) is absorbed either by conversion of adjacent silicate rock into carbonate rock or by conversion of carbonate rock into water soluble bicarbonate. In both cases natural gas, which is primarily methane (CH4), is left behind.
OIL:
At temperatures in the range 20 degrees C to 40 degrees C there are multiple natural anaerobic carbohydrate biochemical decomposition reactions of the form:
C6H12O6 = 2 C2H5OH + 2 CO2
The released CO2 is absorbed by conversion of adjacent silicate rock to carbonate rock leaving behind the ethanol (C2H5OH).
With some heating and further dehydration by an anhydrous volcanic rock such as alumina, C2H5OH reforms into a mixture of heavier alcohols. eg:
2 C2H5OH = C4H9OH + H2O
C2H5OH + C4H9OH = C6H13OH + H2O
2 C4H9OH = C8H17OH + H2O
C4H9OH + C8H17OH = C12H25OH + H2O
The heavier alcohols then slowly combine with adjacent coal (C) to form a mixture of oils plus CO2. eg
2 C6H13OH + C = 2 C6H14 + CO2
2 C8H17OH + C = 2 C8H18 + CO2
2 C12H25OH + C = 2 C12H26 + CO2
The released carbon dioxide (CO2) is absorbed by further conversion of silicate rock into carbonate rock or by conversion of carbonate rock into water soluble bicarbonate leaving behind a mixture of oils known as petroleum.
THE EFFECTS OF MANKIND:
Commencing with the industrial revolution, mankind's extraction and combustion of fossil fuels (coal, oil and natural gas) reversed the natural evolutionary trend of gradual movement of carbon from Pool #1 to Pool #2. Combustion of fossil fuels causes rapid chemical reactions of the form:
Coal:
C + O2 = CO2
Natural Gas:
CH4 + 2 (O2) = CO2 + 2 (H2O)
Oil:
N (CH2) + N (3/ 2)(O2) = N (CO2) + N (H2O)
that all produce carbon dioxide (CO2) which is released to the atmosphere.
Part of the resulting extra non-equilibrium atmospheric CO2 has been absorbed by the oceans via conversion of exposed insoluble marine metal carbonates such as CaCO3 into water soluble bicarbonate compounds such as Ca(HCO3)2 . However, the balance of the extra CO2 remains in the atmosphere. The measured increase in atmospheric CO2 concentration from its preindustrial value of .028% to its present value of .040% has significantly reduced thermal infrared energy emission from the Earth. The resulting ongoing heat retention by the Earth has melted the floating Arctic ice and is warming the oceans.
During the 20th century the rate of rise of average sea level tripled from 1.06 mm / year to 3.18 mm / year and there was substantial acidification of the oceans due to addition of bicarbonate (HCO3)- ions and depletion of exposed marine metal carbonates. With an increasing atmospheric CO2 concentration heat is increasingly transported upwards through the atmosphere via convection instead of via radiation. This effect together with warming of the ocean surface has increased the frequency and intensity of major storms.
Warmer average winter temperatures have caused loss of mountain glaciers for supply of summer agricultural irrigation, have caused massive damage to Canadian forests via insect infestation and have reduced the working life of winter ice roads.
The increased atmospheric CO2 concentration has lowered the pH of rain water, which is causing accelerated corrosion and deterioration of limestone, concrete and steel based structures that are exposed to rain water including but not limited to: marble monuments, marble stairs, marble building facing, bridges, pipes, building balconies, railings, roadways, railway track, electricity transmission towers, man holes, sewer grates and utility poles.
WARMING CYCLE ENTRAPMENT:
The present rate of injection of fossil CO2 into the Earth's atmosphere greatly exceeds the increased rate of CO2 absorption by the ocean due to melting of the Arctic Ocean ice cover, so the Earth is trapped in a warming cycle from which it cannot escape as long as massive combustion of fossil fuels continues.
This web page last updated March 18, 2017.
| Home | Energy | Nuclear Fusion | Electricity | Climate Change | Lighting Control | Contacts | Links |
|---|